Analyzing RASSF1A Promoter Methylation and MicroRNA Profiles in the Plasma of Breast Cancer Patients from Pakistan
Abstract
 Abstract Views: 18
Abstract Views: 18
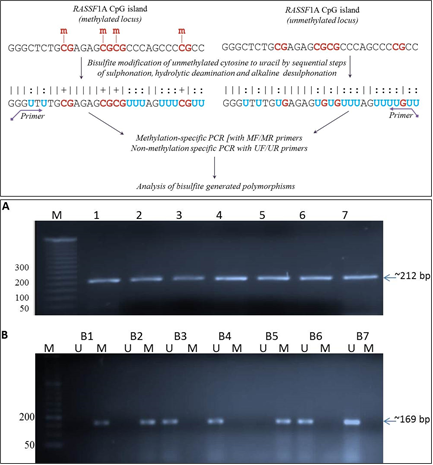
The current study assessed the promoter DNA methylation status of a tumor suppressor gene, namely Ras association domain family 1 isoform A (RASSF1A), and the expression levels of selected cancer-associated microRNAs (miR-10b and miR-34a) in plasma samples of breast cancer patients, with the aim of evaluating the diagnostic relevance of circulating nucleic acids. Circulating cell free DNA (cfDNA) was isolated from the plasma samples of 40 breast cancer patients, followed by bisulfite modification and methylation-specific PCR to determine RASSF1A promoter hypermethylation. Further, miR-specific real-time qRT-PCR approach was used to analyse the levels of microRNA employing DNA primers, instead of stem-loop/locked nucleic acid (LNA) primers. RASSF1A promoter hypermethylation was detected in 40% of patients. Circulating miR-10b levels showed a statistically significant difference between patients and healthy controls (p < 0.001). Receiver operating characteristic (ROC) curve analysis demonstrated a high discriminative performance of miR-10b, with an area under the curve (AUC) of 0.97 (95% CI: 0.90–0.99; SE = 0.16). The results point to the potential value of circulating microRNA profiling, especially miR-10b, as an affordable and invasive supplement to the current breast cancer detection techniques. However, more validation research in a larger cohort is required to prove their therapeutic value.
Downloads
References
Zaib S, Masood N, Khan JS, Yasmin A. Breast cancer: epidemiology, risk factors and survival analysis in the Pakistani population. J Univ Coll Med Dent. 2024;4:18-23. https://doi.org/10.51846/jucmd.v3i1.2784
Zaheer S, Shah N, Maqbool SA, Soomro NM. Estimates of past and future time trends in age-specific breast cancer incidence among women in Karachi, Pakistan: 2004–2025. BMC Public Health. 2019;19(1):e1001. https://doi.org/10.1186/s12889-019-7330-z
Cardinali B, Tasso R, Piccioli P, Ciferri MC, Quarto R, Del Mastro L. Circulating miRNAs in breast cancer diagnosis and prognosis. Cancers (Basel). 2022;14(9):e2317. https://doi.org/10.3390/cancers14092317
Szczepanek J, Skorupa M, Jarkiewicz-Tretyn J, Cybulski C, Tretyn A. Harnessing epigenetics for breast cancer therapy: the role of DNA methylation, histone modifications, and microRNA. Int J Mol Sci. 2023;24(8):e7235. https://doi.org/10.3390/ijms24087235
Prabhu KS, Sadida HQ, Kuttikrishnan S, Junejo K, Bhat AA, Uddin S. Beyond genetics: exploring the role of epigenetic alterations in breast cancer. Pathol Res Pract. 2024;254:e155174. https://doi.org/10.1016/j.prp.2024.155174
Gonzalez T, Nie Q, Chaudhary LN, Basel D, Reddi HV. Methylation signatures as biomarkers for non-invasive early detection of breast cancer: a systematic review of the literature. Cancer Genet. 2024;282:1-8. https://doi.org/10.1016/j.cancergen.2023.12.003
Rajkumar T, Amritha S, Sridevi V, et al. Identification and validation of plasma biomarkers for diagnosis of breast cancer in South Asian women. Sci Rep. 2022;12(1):e100. https://doi.org/10.1038/s41598-021-04176-w
Panagopoulou M, Esteller M, Chatzaki E. Circulating cell-free DNA in breast cancer: searching for hidden information towards precision medicine. Cancers (Basel). 2021;13(4):e728. https://doi.org/10.3390/cancers13040728
El Hejjioui B, Bouguenouch L, Melhouf MA, El Mouhi H, Bennis S. Clinical evidence of circulating tumor DNA application in aggressive breast cancer. Diagnostics (Basel). 2023;13(3):e470. https://doi.org/10.3390/diagnostics13030470
Vlataki K, Antonouli S, Kalyvioti C, et al. Circulating tumor DNA in the management of early-stage breast cancer. Cells. 2023;12(12):e1573. https://doi.org/10.3390/cells12121573
Gupta V, Agarwal P, Deshpande P. Impact of RASSF1A gene methylation on clinico-pathological features of tumor and non-tumor tissue of breast cancer. Ann Diagn Pathol. 2021;52:e151722. https://doi.org/10.1016/j.anndiagpath.2021.151722
Vietri MT, Delia G, Benincasa G, Ferraro G, Caliendo G, Nicoletti GF, et al. DNA methylation and breast cancer: a way forward. Int J Oncol. 2021;59(5):1-2. https://doi.org/10.3892/ijo.2021.5278
Hum M, Lee AS. DNA methylation in breast cancer: early detection and biomarker discovery through current and emerging approaches. J Transl Med. 2025;23(1):e465. https://doi.org/10.1186/s12967-025-06495-2
Zhang X, Zhao D, Yin Y, et al. Circulating cell-free DNA-based methylation patterns for breast cancer diagnosis. NPJ Breast Cancer. 2021;7(1):e106. https://doi.org/10.1038/s41523-021-00316-7
Cao YN, Li QZ, Liu YX, Jin W, Hou R. Discovering the key genes and important DNA methylation regions in breast cancer. Hereditas. 2022;159(1):e7. https://doi.org/10.1186/s41065-022-00220-5
Arif T, Anwar N. Promoter hypermethylation and expression changes of BRCA1 gene in a cohort of sporadic breast cancer cases among Pakistani population. Asian Pac J Cancer Prev. 2020;21(8):2395-2401. https://doi.org/10.31557/APJCP.2020.21.8.2395
He S, Hou Y, Hou L, et al. Targeted RASSF1A expression inhibits proliferation of HER2-positive breast cancer cells in vitro. Exp Ther Med. 2023;25(6):e245. https://doi.org/10.3892/etm.2023.11944
Bin Y, Ding Y, Xiao W, Liao A. RASSF1A: a promising target for the diagnosis and treatment of cancer. Clin Chim Acta. 2020;504:98-108. https://doi.org/10.1016/j.cca.2020.01.014
Xin-Ling D, Chao-Si Q, Gui-Ting T, Xue-Yan C, Li-Ying F, Ying-Hua Y. Association of methylation of RASSF1A between breast cancer and benign breast lesions: a meta-analysis. Clin Surg. 2020;5:e2789.
Garcia-Martinez L, Zhang Y, Nakata Y, Chan HL, Morey L. Epigenetic mechanisms in breast cancer therapy and resistance. Nat Commun. 2021;12(1):1786. https://doi.org/10.1038/s41467-021-22024-3
Uzuner E, Ulu GT, Gürler SB, Baran Y. The role of miRNA in cancer: pathogenesis, diagnosis, and treatment. In: Allmer J, Yousef M, eds. miRNomics. Methods in Molecular Biology. Vol 2257. Humana; 2022. https://doi.org/10.1007/978-1-0716-1170-8_18
Otmani K, Lewalle P. Tumor suppressor miRNA in cancer cells and the tumor microenvironment: mechanism of deregulation and clinical implications. Front Oncol. 2021;11:e708765. https://doi.org/10.3389/fonc.2021.708765
Kanwal N, Al Samarrai O, Al-Zaidi HM, Mirzaei A, Heidari M. Comprehensive analysis of microRNA (miRNA) in cancer cells. Cell Mol Biomed Rep. 2023;3(2):89-97.
Imran K, Iqbal MJ, Ahmed MM, et al. Epigenetic dysregulation in cancer: mechanisms, diagnostic biomarkers and therapeutic strategies. Med Oncol. 2025;42(8):e359. https://doi.org/10.1007/s12032-025-02905-z
Beilankouhi EA, Maghsoodi MS, Sani MZ, et al. miRNAs that regulate apoptosis in breast cancer and cervical cancer. Cell Biochem Biophys. 2024;82(3):1993-2006. https://doi.org/10.1007/s12013-024-01405-7
Xing Y, Ruan G, Ni H, et al. Tumor immune microenvironment and its related miRNAs in tumor progression. Front Immunol. 2021;12:e624725. https://doi.org/10.3389/fimmu.2021.624725
McGuire A, Casey MC, Waldron RM, et al. Prospective assessment of systemic microRNAs as markers of response to neoadjuvant chemotherapy in breast cancer. Cancers (Basel). 2020;12(7):e1820. https://doi.org/10.3390/cancers12071820
Halim A, Kim B, Kenyon E, Moore A. miR-10b as a clinical marker and a therapeutic target for metastatic breast cancer. Technol Cancer Res Treat. 2025;24:1-7. https://doi.org/10.1177/15330338251339256
Asadirad A, Khodadadi A, Talaiezadeh A, et al. Evaluation of miRNA-21-5p and miRNA-10b-5p levels in serum-derived exosomes of breast cancer patients in different grades. Mol Cell Probes. 2022;64:e101831. https://doi.org/10.1016/j.mcp.2022.101831
Elgeshy KM, Abdel Wahab AH. The role, significance, and association of microRNA-10a/b in physiology of cancer. MicroRNA. 2022;11(2):118-138. https://doi.org/10.2174/2211536611666220523104408
Yoo B, Ross A, Pantazopoulos P, Medarova Z. miRNA10b-directed nanotherapy effectively targets brain metastases from breast cancer. Sci Rep. 2021;11(1):e2844. https://doi.org/10.1038/s41598-021-82528-2
Halim A, Al-Qadi N, Kenyon E, et al. Inhibition of miR-10b treats metastatic breast cancer by targeting stem cell-like properties. Oncotarget. 2024;15:591-606. https://doi.org/10.18632/oncotarget.28641
Tokumaru Y, Katsuta E, Oshi M, et al. High expression of miR-34a associated with less aggressive cancer biology but not with survival in breast cancer. Int J Mol Sci. 2020;21(9):e3045. https://doi.org/10.3390/ijms21093045
Li S, Wei X, He J, et al. The comprehensive landscape of miR-34a in cancer research. Cancer Metastasis Rev. 2021;40(3):925-948. https://doi.org/10.1007/s10555-021-09973-3
Li W, Wang Y, Liu R, et al. MicroRNA-34a: potent tumor suppressor, cancer stem cell inhibitor, and potential anticancer therapeutic. Front Cell Dev Biol. 2021;9:e640587. https://doi.org/10.3389/fcell.2021.640587
Copyright (c) 2025 Jawaria Shaheen, Safia Firdous, Khulod Ibrahim Hassan, Saima Sadaf

This work is licensed under a Creative Commons Attribution 4.0 International License.
Author(s) retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution (CC-BY) 4.0 License that allows others to share the work with an acknowledgment of the work’s authorship and initial publication in this journal.






