The Optimization of the Breeding Conditions of Aspergillus niger for En-zyme Production Focusing on Temperature, Substrate Source, Nitro-gen Source, Carbon Source, and pH
Abstract
 Abstract Views: 116
Abstract Views: 116
Background. Aspergillus niger is extensively employed in the industry to produce organic acids, enzymes, and other metabolites. Optimizing the breeding conditions for A. niger to increase enzyme yields and boost the effectiveness of industrial fermentation processes is a crucial field of study.
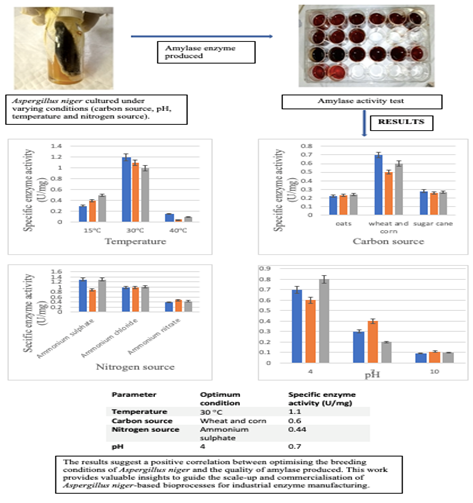
Methods The effects of different temperature conditions (15°C, 30°C, and 45°C), carbon sources (oats, wheat, corn, and sugar cane), nitrogen sources (NH4NO3, NH4CL, and (NH4)2SO4), and pH (4, 7, and 10) on the production of enzyme amylase were determined. A. niger was fermented under controlled conditions in bioreactor shake flasks and enzyme activities were measured using standard colorimetric and fluorometric assays.
Results. The results showed an optimum temperature of 30°C for A. niger fermentation for the production of amylase, with the highest specific enzyme activity of 1.1 U/mg. NH4SO4 was found to be the best nitrogen source for the fermentation of A. niger for enzyme production. Whereas, wheat and corn proved to be the best carbon sources for the fermentation of A. niger for enzyme production. Wheat and corn produced amylase with the specific enzyme activity of 0.63 U/mg. The pH 4 was found to be ideal for the fermentation of A. niger for enzyme production The enzyme produced at pH 4 showed a specific enzyme activity of 0.5 U/mg.
Conclusion. The results suggest a positive correlation between optimising the breeding conditions of A. niger and the quality of amylase produced. It provides valuable insights to guide the scale-up and commercialization of A. niger-based bioprocesses for industrial enzyme manufacturing.
Downloads
References
Simoni R, Hill R, Vaughan M. Urease, the first crystalline enzyme and the proof that enzymes are proteins: the work of James B. Sumner. J Biol Chem. 2022;277(35):1-2. https://doi.org/10.1016/S0021-9258(20)69970-7 DOI: https://doi.org/10.1016/S0021-9258(20)69970-7
Sharma A, Arya S. Biocatalysis as a green approach for industrial waste treatment. Biomolecules. 2020;10:359-405. https://doi.org/10.1007/978-3-030-44176-0_14 DOI: https://doi.org/10.1007/978-3-030-44176-0_14
Mokrani S, Nabti E. Recent status in production, biotechnological applications, commercial aspects, and future prospects of microbial enzymes: a comprehensive review. Int J Agric Sci Food Technol. 2024;6:6-20.
Yadav D, Chowdary P, Anand G, Gaur R. Microbial enzymes: production, purification and industrial applications. In: Yadav D, Chowdhary P, Anand G, Gau RK, eds. Microbial Enzymes. Wiley; 2025:38-48.
https://doi.org/10.1002/9783527844340 DOI: https://doi.org/10.1002/9783527844340
Nguyen H. Immobilised enzymes in biosensor applications. Materials. 2019;12(1):e121.
https://doi.org/10.3390/ma12010121 DOI: https://doi.org/10.3390/ma12010121
Tate A, Sahu L, Pandey J, Mishra M, Sharma P. Green catalysis for chemical transformation: the need for sustainable development. Curr Res Green Sustain Chem. 2022;3:122-141.
https://doi.org/10.1016/j.crgsc.2021.100248 DOI: https://doi.org/10.1016/j.crgsc.2021.100248
Popielarz D, Farkas P, Bzducha-Wróbel A. Current directions of selected plant-origin wastes valorization in biotechnology of food additives and other important chemicals. Foods. 2025;14:e954.
https://doi.org/10.3390/foods14060954 DOI: https://doi.org/10.3390/foods14060954
Lu D, Zhang M, Wang F, et al. Nutritional value improvement of soybean meal through solid-state fermentation by proteases-enhanced Streptomyces sp. SCUT-3. Int J Biol Macromol. 2025;298:e140035.
https://doi.org/10.1016/j.ijbiomac.2025.140035 DOI: https://doi.org/10.1016/j.ijbiomac.2025.140035
Arnau J, Yaver D, Hjort C. Strategies and challenges for the development of industrial enzymes using fungal cell factories. Biotechnol Adv. 2020;38:107-118. https://doi.org/10.1007/978-3-030-29541-7_7 DOI: https://doi.org/10.1007/978-3-030-29541-7_7
Santos-Beneit F. What is the role of microbial biotechnology and genetic engineering in medicine? MicrobiologyOpen. 2024;13(2):e1234. https://doi.org/10.1002/mbo3.1406 DOI: https://doi.org/10.1002/mbo3.1406
Yafetto L. Application of solid-state fermentation for bioprocessing of agro-industrial wastes: a bibliometric analysis. Heliyon. 2022;8:e10412. https://doi.org/10.1016/j.heliyon.2022.e09173 DOI: https://doi.org/10.1016/j.heliyon.2022.e09173
Kabir M, Ovi A, Ju L. Real-time pH and temperature monitoring in solid-state fermentation reveals culture physiology and optimizes enzyme harvesting. Microb Cell Fact. 2025;24:e188.
https://doi.org/10.1186/s12934-025-02820-y DOI: https://doi.org/10.1186/s12934-025-02820-y
Li Z, Kong H, Li Z, Gu Z, Ban X, Hong Y, et al. Designing liquefaction and saccharification processes of highly concentrated starch slurry. Compr Rev Food Sci Food Saf. 2023;22:1597-1612.
https://doi.org/10.1111/1541-4337.13122 DOI: https://doi.org/10.1111/1541-4337.13122
Kim J, Kim M, Yim J, Kim I, Rhee J, Han S. Isolation and characterization of low-temperature and high-salinity amylase from Halomonas sp. KS41843. Fermentation. 2025;11:e465.
https://doi.org/10.3390/fermentation11080465 DOI: https://doi.org/10.3390/fermentation11080465
Gomez E, Delgado J, Gonzalez J. Influence of water availability on estimates of microbial extracellular enzyme activity. PeerJ. 2021;9:e10994. https://doi.org/10.7717/peerj.10994 DOI: https://doi.org/10.7717/peerj.10994
Jamilu H, Ibrahim A, Abdullahi S. Isolation, optimization and characterization of lipase-producing bacteria from abattoir soil. Int J Sci Adv. 2022;3(1):75-82. https://doi.org/10.51542/ijscia.v3i1.9 DOI: https://doi.org/10.51542/ijscia.v3i1.9
Gu S, Chen Z, Wang F, Wang X. Characterization and inhibition of four fungi producing citrinin in various culture media. Biotechnol Lett. 2021;43:701-710. https://doi.org/10.1007/s10529-020-03061-2 DOI: https://doi.org/10.1007/s10529-020-03061-2
Raveendran S, Parameswaran B, Sabeela B, Abraham A, Mathew A. Applications of microbial enzymes in the food industry. Food Technol Biotechnol. 2018;56(1):16-30. https:// doi.org/10.17113/ftb.56.01.18.5491
Kaur A, Rishi V, Soni S, Rishi P. A novel multi-enzyme preparation produced from Aspergillus niger using biodegradable waste. AMB Express. 2020;10:e36. https://doi.org/10.1186/s13568-020-00970-3 DOI: https://doi.org/10.1186/s13568-020-00970-3
Makeri MS, Bala M, Wante SP, Bitrus KV, Aliyu HU. Activity of α-amylase produced by Aspergillus niger under different conditions using solid-state fermentation. Asian J Biotechnol Bioresour Technol. 2021;7:1-11.
https://doi.org/10.9734/ajb2t/2021/v7i230095 DOI: https://doi.org/10.9734/ajb2t/2021/v7i230095
Ramasamy S. Utilizing aerobic and anaerobic mesophilic fungi for production of multienzymes. In: Uppuluri KB, Selvasembian R. eds. Bioprospecting of Multi-tasking for a Sustainable Environment. Springer; 2024:49-67.
https://doi.org/10.1007/978-981-97-4113-7_3 DOI: https://doi.org/10.1007/978-981-97-4113-7_3
Thuy N, Coleman T, Christopher M, et al. Extremely acidophilic filamentous fungi are more prevalent in diverse ecosystems than previously documented. Sci Rep. 2025;15:e30445. https://doi.org/10.1038/s41598-025-06321-1 DOI: https://doi.org/10.1038/s41598-025-06321-1
Farh M, Abdellaoui N, Seo J. pH changes affect gene expression and hydrolytic enzyme production in Saccharomyces fibuligera. Front Microbiol. 2021;12:1-10. https://doi.org/10.3389/fmicb.2021.672661 DOI: https://doi.org/10.3389/fmicb.2021.672661
Schepper C, Michiels P, Buve C, Van Loey A, Courtin C. Starch hydrolysis during mashing: activity and thermal inactivation kinetics of barley malt amylases. Carbohydr Polym. 2021;256:117-125.
https://doi.org/10.1016/j.carbpol.2020.117494 DOI: https://doi.org/10.1016/j.carbpol.2020.117494
Freitas M, Proença C, Ribeiro D, Quinaz-Garcia M, Araújo A, Fernandes E. Assessment of α-amylase activity in a microanalysis system. J Chem Educ. 2023;100(3):1237-1245. https://doi.org/10.1021/acs.jchemed.2c00392 DOI: https://doi.org/10.1021/acs.jchemed.2c00392
Bellaouchi R, Abouloifa H, Rokni Y, et al. Characterization and optimization of extracellular enzyme production by Aspergillus niger. J Genet Eng Biotechnol. 2021;19(1):e50.
https://doi.org/10.1186/s43141-021-00145-y DOI: https://doi.org/10.1186/s43141-021-00145-y
Adegbanke A, Adepomola A, Adeniran E, Bamidele O. Effect of pH and temperature on isolation and characterization of α-amylase from Aspergillus niger. Acta Sci Nutr Health. 2021;5(12):1-10.
Copyright (c) 2026 Rumbidzai Mangoyi, Tanaka Madingi, Marcia Matongorere

This work is licensed under a Creative Commons Attribution 4.0 International License.
BSR follows an open-access publishing policy and full text of all published articles is available free, immediately upon publication of an issue. The journal’s contents are published and distributed under the terms of the Creative Commons Attribution 4.0 International (CC-BY 4.0) license. Thus, the work submitted to the journal implies that it is original, unpublished work of the authors (neither published previously nor accepted/under consideration for publication elsewhere). On acceptance of a manuscript for publication, a corresponding author on the behalf of all co-authors of the manuscript will sign and submit a completed the Copyright and Author Consent Form.














