Advances in Understanding Candida Species: Mechanisms of Antifungal Resistance and Emerging Therapies
Abstract
 Abstract Views: 331
Abstract Views: 331
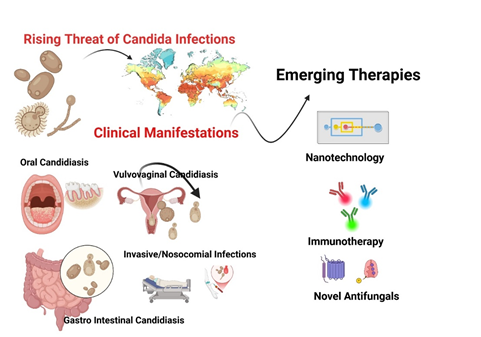
Background. The rising incidence of fungal infections and the growing threat of antifungal resistance have heightened the clinical importance of medical mycology. Candida species, particularly Candida albicans, are major contributors to fungal diseases, including vulvovaginal and oral candidiasis.
Methods. This mini review critically analyzed the current literature on Candida infection, primarily Candida albicans, growing antifungal resistance, and therapeutic limitations. Furthermore, this review also explored the emerging therapeutic strategies, including novel antifungal agents and nanotechnology-based strategies to overcome biofilm-related resistance.
Results. Recent studies highlight a rising prevalence of non-albicans Candida species (NAC), many of which exhibit increased antifungal resistance and a tendency for persistent infections. Nevertheless, Candida albicans remains a major pathogenic species, capable of evading the host immune response through robust biofilm formation and other virulence factors.
Conclusion. Infections caused by Candida species pose a significant healthcare challenge due to their rising incidence and increasing resistance to conventional antifungals. The complexity of the infection(s) is due to C.albicans accompanied by non-albicans species. This review emphasized on urgent need of emerging therapeutic strategies for biofilm-associated resistance.
Downloads
References
Bajpai VK, Khan I, Shukla S, et al. Invasive fungal infections and their epidemiology: measures in the clinical scenario. Biotechnol Biopro Eng. 2019;24:436–444. https://doi.org/ 10.1007/s12257-018-0477-0 DOI: https://doi.org/10.1007/s12257-018-0477-0
Chavez-Bueno S, McCulloh RJ. Current trends in epidemiology and antimicrobial resistance in neonatal sepsis. In: Vincent J-L, ed. Annual Update in Intensive Care and Emergency Medicine 2018. Springer Nature; 2018:39–51. DOI: https://doi.org/10.1007/978-3-319-73670-9_4
Pinho S, Miranda IM, Costa-de-Oliveira S. Global epidemiology of invasive infections by uncommon candida species: a systematic review. J Fungi. 2024;10(8):e558. https://doi.org/10.3390/jof10080558
Vila T, Sultan AS, Montelongo-Jauregui D, Jabra-Rizk MA. Oral candidiasis: a disease of opportunity. J Fungi. 2020;6(1):e15. https://doi.org /10.3390/jof6010015 DOI: https://doi.org/10.3390/jof6010015
Firoozi P, Farshidfar N, Fekrazad R. Efficacy of antimicrobial photodynamic therapy compared to nystatin therapy in reducing Candida colony count in patients with Candida-associated denture stomatitis: a systematic review and meta-analysis. Evid Based Dent. 2021:1–8. https://doi.org/10.1038/s41432-021-0208-9 DOI: https://doi.org/10.1038/s41432-021-0208-9
Nsenga L, Bongomin F. Recurrent candida vulvovaginitis. Venereology. 2022;1(1):114–123. https://doi.org/ 10.3390/venereology1010008
Xiao M, Fan X, Chen SCA, et al. Antifungal susceptibilities of Candida glabrata species complex, Candida krusei, Candida parapsilosis species complex and Candida tropicalis causing invasive candidiasis in China: 3 year national surveillance. J Antimicrobl Chem. 2015;70(3):802–810. https://doi.org/10.1093/jac/ dku460 DOI: https://doi.org/10.1093/jac/dku460
Mba IE, Nweze EI. Mechanism of Candida pathogenesis: revisiting the vital drivers. Eur J Clinic Microbiol Infect Dis. 2020;39(10):1797–1819. https://doi.org/10.1007/s10096-020-03912-w DOI: https://doi.org/10.1007/s10096-020-03912-w
Gerard R, Sendid B, Colombel J-F, Poulain D, Jouault T. An immunological link between Candida albicans colonization and Crohn’s disease. Eur J Clin Microbiol Infect Dis. 2015;41(2):135–139. https://doi.org/10.3109/1040841X.2013.810587 DOI: https://doi.org/10.3109/1040841X.2013.810587
Wang H, Wu H, Li K-D, et al. Intestinal fungi and systemic autoimmune diseases. Autoimmun Rev. 2023;22(2):e103234. https://doi.org /10.1016/j.autrev.2022.103234 DOI: https://doi.org/10.1016/j.autrev.2022.103234
Jangi S, Hsia K, Zhao N, et al. Dynamics of the gut mycobiome in patients with ulcerative colitis. Clinic Gastroenterol Hepatol. 2024;22(4):821–830. https://doi.org /10.1016/j.cgh.2023.09.023 DOI: https://doi.org/10.1016/j.cgh.2023.09.023
Gutierrez MW, van Tilburg Bernardes E, Changirwa D, McDonald B, Arrieta M-C. “Molding” immunity—modulation of mucosal and systemic immunity by the intestinal mycobiome in health and disease. Mucosal Immunol. 2022;15(4):573–583. https://doi.org/10.1038/s41385-022-00515-w DOI: https://doi.org/10.1038/s41385-022-00515-w
Alonso-Monge R, Gresnigt MS, Román E, Hube B, Pla J. Candida albicans colonization of the gastrointestinal tract: a double-edged sword. Plos Pathog. 2021;17(7):e1009710. https://doi.org/ 10.1371/journal.ppat.1009710 DOI: https://doi.org/10.1371/journal.ppat.1009710
Lass-Flörl C, Kanj SS, Govender NP, Thompson III GR, Ostrosky-Zeichner L, Govrins MA. Invasive candidiasis. Nat Rev Dis Primers. 2024;10(1):e20. https://doi.org/10.1038/s41572-024-00503-3 DOI: https://doi.org/10.1038/s41572-024-00503-3
Chahine A, Farooqi S, Marzvanyan A, Michael M, Chavez-Frazier A, Guzman N. Mucocutaneous Candida infections in immunocompromised patients. Curr Derm Rep. 2022;11(2):73–88. https://doi.org/ 10.1007/s13671-022-00356-7 DOI: https://doi.org/10.1007/s13671-022-00356-7
Olum R, Baluku JB, Okidi R, Andia-Biraro I, Bongomin F. Prevalence of HIV-associated esophageal candidiasis in sub-Saharan Africa: a systematic review and meta-analysis. Tropical Medicine and Health. 2020;48:e82. https://doi.org/10.1186/ s41182-020-00268-x DOI: https://doi.org/10.1186/s41182-020-00268-x
Carpintieri S, Uyar E, Buryk Y. Opportunistic infections and malignancies in a patient with HIV/AIDS and a critically Low CD4 Count of 1 Cell/μL. Cureus. 2024;16(5):e60129. https://doi.org/ 10.7759/cureus.60129 DOI: https://doi.org/10.7759/cureus.60129
Ucar S, Naik AQ, Zafar T. Cancer and fungal infections. In: Masood N, Yasmin A, eds. The Microbiome And Cancer: Understanding the Role of Microorganisms in Tumor Development And Treatment. Springer Nature; 2024:209–226. DOI: https://doi.org/10.1007/978-981-97-6948-3_9
Caruso R, Lo BC, Chen GY, Núñez G. Host–pathobiont interactions in Crohn’s disease. Nat Rev Gastroenterol Hepatol. 2025;(6):395–414. https://doi.org/10.1038/s41575-024-00997-y DOI: https://doi.org/10.1038/s41575-024-00997-y
Gedefie A, Shimeles G, Motbainor H, Kassanew B, Genet C. Vaginal colonization and vertical transmission of Candida species: prevalence and associated factors among pregnant women and their neonates at public health facilities of Northeast Ethiopia. BMC Pregnan Child. 2025;25(1):e22. https://doi.org/10.1186/s12884-024-07103-9 DOI: https://doi.org/10.1186/s12884-024-07103-9
Nagendra L, Mondal S, Bhattacharya S. Thyroid Disorders in the Tropics. Endotext; 2024.
Ikuta KS, Meštrović T, Naghavi M. Global incidence and mortality of severe fungal disease. Lancet Infect Dis. 2024;24(5):e268. https://doi.org/ 10.1016/s1473-3099(24)00102-6 DOI: https://doi.org/10.1016/S1473-3099(24)00102-6
El-Ganiny AM, Yossef NE, Kamel HA. Prevalence and antifungal drug resistance of nosocomial Candida species isolated from two university hospitals in Egypt. Current Med Mycol. 2021;7(1):e31. https://doi.org/ 10.1016/s1473-3099(24)00102-6 DOI: https://doi.org/10.18502/cmm.7.1.6181
Aydin S, Derin O, Sahin M, et al. Epidemiology of nosocomial candidemia, mortality, and antifungal resistance: 7-year experience in Turkey. Jpn J Infect Dis. 2022;75(6):597–603. DOI: https://doi.org/10.7883/yoken.JJID.2022.181
Prajapati J. Antifungal susceptibility and speciation of Candida isolated from blood at a tertiary care centre. Int J Med Med Res. 2024;10(1):32–39. DOI: https://doi.org/10.61751/ijmmr/1.2024.32
Dutta A. Candidiasis: the laboratory report states that there are yeast in the blood culture! In: Domachowske J, ed. Introduction to Clinical Infectious Diseases: A Problem-Based Approach. Springer; 2025:487–493. DOI: https://doi.org/10.1007/978-3-031-84089-0_44
Katsipoulaki M, Stappers MH, Malavia-Jones D, et al. Candida albicans and Candida glabrata: global priority pathogens. Microbiol Molecul Biol Rev. 2024;88(2):e00021-23. https://doi.org/10.1128/mmbr.00021-23.
Malavika G, Ravi SSS, Maheswary D, Leela K, Lathakumari RH, Priya KSL. Role of Candida albicans in chronic inflammation and the development of oral squamous cell carcinoma. Cancer Pathog Ther. 2025. https://doi.org/ 10.1016/j.cpt.2025.03.002
Casu C, Pinna M, Denotti G, Murgia M, Orru G. Association between C. albicans and leukoplakia and its treatment with photodynamic therapy: a review of the literature and a case report. World Cancer Res J. 2023;10:e2712. https://dx.doi.org/ 10.32113/wcrj_202312_2712
Tasso CO, Ferrisse TM, de Oliveira AB, Ribas BR, Jorge JH. Candida species as potential risk factors for oral squamous cell carcinoma: systematic review and meta-analysis. Cancer Epidemiol. 2023;86:e102451. https://doi.org/10.1016/j.canep.2023.102451 DOI: https://doi.org/10.1016/j.canep.2023.102451
Ayuningtyas NF, Mahdani FY, Pasaribu TAS, et al. Role of Candida albicans in oral carcinogenesis. Pathophysiology. 2022;29(4):650–662. https://doi.org/10.3390/ pathophysiology29040051 DOI: https://doi.org/10.3390/pathophysiology29040051
Yang Z, Zhang S, Ji N, Li J, Chen Q. The evil companion of OSCC: Candida albicans. 2024;30(4):1873–1886.https://doi.org/10.1111/odi.14700 DOI: https://doi.org/10.1111/odi.14700
Abuhajar E, Ali K, Zulfiqar G, et al. Management of chronic atrophic candidiasis (denture stomatitis)—a narrative review. Int J Environ Res Public Health. 2023;20(4):e3029. https://doi.org/10.3390/ijerph20043029 DOI: https://doi.org/10.3390/ijerph20043029
Katsipoulaki M, Stappers MH, Malavia-Jones D, Brunke S, Hube B, Gow NA. Candida albicans and Candida glabrata: global priority pathogens. Microbiol Molecul Biol Rev. 2024;88(2):e00021-23. https:// doi.org/10.1128/mmbr.00021-23 DOI: https://doi.org/10.1128/mmbr.00021-23
Satora M, Grunwald A, Zaremba B, et al. Treatment of vulvovaginal candidiasis—an overview of guidelines and the latest treatment methods. J Clinic Med. 2023;12(16):e5376. https://doi.org/10.3390/jcm12165376
Benedict K, Singleton AL, Jackson BR, Molinari NAM. Survey of incidence, lifetime prevalence, and treatment of self-reported vulvovaginal candidiasis, United States, 2020. BMC Women's Health. 2022;22(1):e147. https://doi.org/10. 1186/s12905-022-01741-x
Hösükoğlu FG, Ekşi F, Erinmez M, Uğur MG. An epidemiologic analysis of vulvovaginal candidiasis and antifungal susceptibilities. Infect Microbe Dis. 2022;4(3):131–136. https://doi.org/10.1097/IM9.0000000000000095 DOI: https://doi.org/10.1097/IM9.0000000000000095
Shahabudin S, Azmi NS, Lani MN, Mukhtar M, Hossain MS. Candida albicans skin infection in diabetic patients: an updated review of pathogenesis and management. Mycoses. 2024;67(6):e13753. https://doi.org/10.1111/myc.13753 DOI: https://doi.org/10.1111/myc.13753
Satora M, Grunwald A, Zaremba B, et al. Treatment of vulvovaginal candidiasis—an overview of guidelines and the latest treatment methods. J Clin Med. 2023;12(16):e5376. https://doi.org/10.3390/jcm12165376 DOI: https://doi.org/10.3390/jcm12165376
Pinho S, Miranda IM, Costa-de-Oliveira S. Global epidemiology of invasive infections by uncommon Candida species: a systematic review. J Fungi. 2024;10(8):e558. https://doi.org/10.3390/jof10080558 DOI: https://doi.org/10.3390/jof10080558
Alves J, Alonso-Tarrés C, Rello J. How to identify invasive candidemia in ICU—a narrative review. Antibiotics. 2022;11(12):e1804. https://doi.org/10.3390/antibiotics11121804 DOI: https://doi.org/10.3390/antibiotics11121804
Pappas PG, Lionakis MS, Arendrup MC, Ostrosky-Zeichner L, Kullberg BJ. Invasive candidiasis. Nat Rev Dis Primers. 2108;4:e18026. https://doi.org/10.1038/nrdp.2018.26 DOI: https://doi.org/10.1038/nrdp.2018.26
Dutta A. Candidiasis: the laboratory report states that there are yeast in the blood culture! In: Domachowske J, ed. Introduction to Clinical Infectious Diseases: A Problem-Based Approach. Springer Nature; 2019:335–340. DOI: https://doi.org/10.1007/978-3-319-91080-2_31
Makled AF, Ali SA, Labeeb AZ, et al. Characterization of Candida species isolated from clinical specimens: insights into virulence traits, antifungal resistance and molecular profiles. BMC Microbiol. 2024;24(1):e388. https://doi.org/10.1186/s12866-024-03515-x DOI: https://doi.org/10.1186/s12866-024-03515-x
Won EJ, Sung H, Kim M-N. Changing epidemiology of clinical isolates of Candida species during the coronavirus disease 2019 pandemic: data analysis from a Korean Tertiary Care Hospital for 6 Years (2017–2022). J Fungi. 2024;10(3):e193. https://doi.org/10.3390/jof10030193 DOI: https://doi.org/10.3390/jof10030193
Liu T, Sun S, Zhu X, Wu H, Sun Z, Peng S. Epidemiology, clinical characteristics, and outcome in candidemia: a retrospective five-year analysis from two tertiary general hospitals. BMC Infect Dis. 2025;25(1):e512. https://doi.org/10. 1186/s12879-025-10908-4 DOI: https://doi.org/10.1186/s12879-025-10908-4
Alenazy H, Alghamdi A, Pinto R, Daneman N. Candida colonization as a predictor of invasive candidiasis in non-neutropenic ICU patients with sepsis: a systematic review and meta-analysis. Int J Infect Dis. 2021;102:357–362. https://doi.org/10. 1016/j.ijid.2020.10.092 DOI: https://doi.org/10.1016/j.ijid.2020.10.092
Soulountsi V, Schizodimos T, Kotoulas SC. Deciphering the epidemiology of invasive candidiasis in the intensive care unit: is it possible? Infection. 2021;49(6):1107–1131. https://doi.org/10.1007/s15010-021-01640-7 DOI: https://doi.org/10.1007/s15010-021-01640-7
Lu S-Y. Oral candidosis: pathophysiology and best practice for diagnosis, classification, and successful management. J Fungi. 2021;7(7):e555. https://doi.org/10. 3390/jof7070555 DOI: https://doi.org/10.3390/jof7070555
Candidiasis P, Candidiasis E, Glossitis MR, et al. Fungal and protozoal infections. In: Color Atlas of Oral and Maxillofacial Diseases. Elsevier Health Sciences; 2018:125–140. DOI: https://doi.org/10.1016/B978-0-323-55225-7.00006-3
Romaneschi N. Effect of a Multispecies Probiotic with Fluconazole on Oral Candidiasis in Subjects with HIV. [master’s thesis]. Connecticut, United States; Yale University; 2024.
Zeise KD, Woods RJ, Huffnagle GB. Interplay between Candida albicans and lactic acid bacteria in the gastrointestinal tract: impact on colonization resistance, microbial carriage, opportunistic infection, and host immunity. Clinic Microbiol. 2021;34(4):e00323-20. https://doi.org /10.1128/CMR.00323-20
Sobel JD. Recurrent vulvovaginal candidiasis. Am J Obstet Gynecol. 2016;214(1):15–21. https://doi.org /10.1016/j.ajog.2015.06.067 DOI: https://doi.org/10.1016/j.ajog.2015.06.067
Nsenga L, Bongomin F. Recurrent candida vulvovaginitis. Venereology. 2022;1(1):114–123. https://doi.org/10.3390/venereology1010008 DOI: https://doi.org/10.3390/venereology1010008
Gonçalves B, Ferreira C, Alves CT, Henriques M, Azeredo J, Silva S. Vulvovaginal candidiasis: epidemiology, microbiology and risk factors. Critic Rev Microbiol. 2016;42(6):905–927. https://doi.org/ 10.3109/1040841X.2015.1091805 DOI: https://doi.org/10.3109/1040841X.2015.1091805
Benedict K, Singleton AL, Jackson BR, Molinari NAM. Survey of incidence, lifetime prevalence, and treatment of self-reported vulvovaginal candidiasis, United States, 2020. BMC Women's Health. 2022;22(1):e147. https://doi.org/10. 1186/s12905-022-01741-x DOI: https://doi.org/10.1186/s12905-022-01741-x
Bojang E, Ghuman H, Kumwenda P, Hall RA. Immune sensing of Candida albicans. J Fungi. 2021;7(2):e119. https://doi.org/10.3390/jof7020119 DOI: https://doi.org/10.3390/jof7020119
Eckstein M-T, Moreno-Velásquez SD, Pérez JC. Gut bacteria shape intestinal microhabitats occupied by the fungus Candida albicans. Curr Biol. 2020;30(23):4799–4807. https://doi. org/10.1016/j.cub.2020.09.027 DOI: https://doi.org/10.1016/j.cub.2020.09.027
Kumamoto CA, Gresnigt MS, Hube B. The gut, the bad and the harmless: Candida albicans as a commensal and opportunistic pathogen in the intestine. Curr Opinion Microbiol. 2020;56:7–15. https://doi.org/10.1016/j.mib. 2020.05.006 DOI: https://doi.org/10.1016/j.mib.2020.05.006
Mochochoko BM, Pohl CH, O’Neill HG. Candida albicans-enteric viral interactions—the prostaglandin E2 connection and host immune responses. iScience. 2023;26(1):e105870. https://doi.org/ 10.1016/j.isci.2022.105870 DOI: https://doi.org/10.1016/j.isci.2022.105870
Sprague JL, Kasper L, Hube B. From intestinal colonization to systemic infections: Candida albicans translocation and dissemination. Gut Micro. 2022;14(1):e2154548. https://doi.org/10.1080/19490976.2022.2154548 DOI: https://doi.org/10.1080/19490976.2022.2154548
Jawhara SJM. How gut bacterial dysbiosis can promote Candida albicans overgrowth during colonic inflammation. Microorganisms. 2022;10(5):e1014. https://doi.org/10. 3390/microorganisms10051014 DOI: https://doi.org/10.3390/microorganisms10051014
Zeise KD, Woods RJ, Huffnagle GB. Interplay between Candida albicans and Lactic Acid Bacteria in the Gastrointestinal Tract: impact on Colonization Resistance, Microbial Carriage, Opportunistic Infection, and Host Immunity. Clinic Microbiol Rev. 2021;34(4):e00323-20. https://doi.org/ 10.1128/CMR.00323-20 DOI: https://doi.org/10.1128/CMR.00323-20
Bratburd JR, Keller C, Vivas E, et al. Gut microbial and metabolic responses to Salmonella enterica serovar typhimurium and Candida albicans. mBio. 2018;9(6):e02032-18. https:// doi.org/10.1128/mbio.02032-18 DOI: https://doi.org/10.1128/mBio.02032-18
Nagaoka I, Tamura H, Reich J. Therapeutic potential of cathelicidin peptide LL-37, an antimicrobial agent, in a murine sepsis model. Int J Mol Sci. 2020;21(17):e5973. https://doi.org/10.3390/ijms21175973 DOI: https://doi.org/10.3390/ijms21175973
Poissy J, Damonti L, Bignon A, et al. Risk factors for candidemia: a prospective matched case-control study. Crit Care. 2020;24:e109. https://doi.org/10.1186/s13054-020-2766-1 DOI: https://doi.org/10.1186/s13054-020-2766-1
Galloway-Peña JR, Kontoyiannis DP. The gut mycobiome: the overlooked constituent of clinical outcomes and treatment complications in patients with cancer and other immunosuppressive conditions. Plos Pathog. 2020;16(4):e1008353. https://doi.org/10.1371/journal.ppat.1008353 DOI: https://doi.org/10.1371/journal.ppat.1008353
Graf K, Last A, Gratz R, et al. Keeping Candida commensal: how lactobacilli antagonize pathogenicity of Candida albicans in an in vitro gut model. Dis Model Mech. 2019;12(9):edmm039719. https://doi.org/10.1242/dmm.039719 DOI: https://doi.org/10.1242/dmm.039719
Tong Y, Tang JJ. Candida albicans infection and intestinal immunity. Microbiol Res. 2017;198:27–35. https://doi.org/10.1016/j.micres.2017.02.002 DOI: https://doi.org/10.1016/j.micres.2017.02.002
Alves R, Barata-Antunes C, Casal M, Brown AJ, Van Dijck P, Paiva S. Adapting to survive: how Candida overcomes host-imposed constraints during human colonization. Plos Pathog. 2020;16(5):e1008478. https://doi.org/10.1371/journal.ppat.1008478 DOI: https://doi.org/10.1371/journal.ppat.1008478
Rana A, Gupta N, Asif S, Thakur A. Surviving the storm: how Candida species master adaptation for pathogenesis. In: Hameed S, Vijayaraghavan P, eds. Recent Advances in Human Fungal Diseases: Progress and Prospects. Springer; 2024:109–155. DOI: https://doi.org/10.1007/978-981-97-4909-6_5
Guinan J, Villa P, Thangamani S. Secondary bile acids inhibit Candida albicans growth and morphogenesis. Pathog Dis. 2018;76(3):efty038. https://doi.org/10.1093/femspd/fty038 DOI: https://doi.org/10.1093/femspd/fty038
Chen H, Zhou X, Ren B, Cheng L. The regulation of hyphae growth in Candida albicans. Virulence. 2020;11(1):337–348. https://doi.org /10.1080/21505594.2020.1748930 DOI: https://doi.org/10.1080/21505594.2020.1748930
Dadar M, Tiwari R, Karthik K, Chakraborty S, Shahali Y, Dhama K. Candida albicans-Biology, molecular characterization, pathogenicity, and advances in diagnosis and control–An update. Microb Pathog. 2018;117:128–138. https://doi.org /10.1016/j.micpath.2018.02.028 DOI: https://doi.org/10.1016/j.micpath.2018.02.028
Ahmad S, Khan ZJ. Invasive candidiasis: a review of nonculture-based laboratory diagnostic methods.Indian J Med Microbiol. 2012;30(3):264–269. https://doi.org /10.4103/0255-0857.99482 DOI: https://doi.org/10.4103/0255-0857.99482
Mikulska M, Calandra T, Sanguinetti M, Poulain D, Viscoli C. The use of mannan antigen and anti-mannan antibodies in the diagnosis of invasive candidiasis: recommendations from the Third European Conference on Infections in Leukemia. Critical Care. 2010;14:1–14. https://doi.org/10.1186/cc9365 DOI: https://doi.org/10.1186/cc9365
Hoenigl M, Enoch DA, Wichmann D, Wyncoll D, Cortegiani A. Exploring European consensus about the remaining treatment challenges and subsequent opportunities to improve the management of invasive fungal infection (IFI) in the Intensive Care Unit. Mycopathologia. 2024;189(3):e41. https://doi.org/ 10.1007/s11046-024-00852-3 DOI: https://doi.org/10.1007/s11046-024-00852-3
Lorenzo-Villegas DL, Gohil NV, Lamo P, et al. Innovative biosensing approaches for swift identification of candida species, intrusive pathogenic organisms. Life. 2023;13(10):e2099. https://doi.org/10.3390/life13102099 DOI: https://doi.org/10.3390/life13102099
Bouza E, Almirante B, García Rodríguez J, et al. Biomarkers of fungal infection: expert opinion on the current situation. Rev Esp Quimioter. 2020;33(1):1–10. https://doi.org /10.37201/req/2260.2019 DOI: https://doi.org/10.37201/req/2260.2019
Allkja J, Roudbary M, Alves AMV, Černáková L, Rodrigues CF. Biomaterials with antifungal strategies to fight oral infections. Critical Rev Biotechnol. 2024;44(6):1151–1163. https://doi.org/10.1080/07388551.2023.2236784 DOI: https://doi.org/10.1080/07388551.2023.2236784
Pereira R, Dos Santos Fontenelle R, De Brito E, de Morais SM. Biofilm of Candida albicans: formation, regulation and resistance. J Appl Microbiol. 2021;131(1):11–22. https://doi.org/10.1111/jam.14949 DOI: https://doi.org/10.1111/jam.14949
Kumar A, Francis A, Hans S, Thakur A. Unravelling drug resistance in Candida species: genetic, biofilm, transcriptional, and epigenetic perspectives. In: Hameed S, Vijayaraghavan P, eds. Recent Advances in Human Fungal Diseases: Progress and Prospects. Springer; 2024:203–237. DOI: https://doi.org/10.1007/978-981-97-4909-6_8
Atriwal T, Azeem K, Husain FM, et al. Mechanistic understanding of Candida albicans biofilm formation and approaches for its inhibition. Front Microbiol. 2021;12:e638609. https://doi.org/10.3389/fmicb.2021.638609 DOI: https://doi.org/10.3389/fmicb.2021.638609
Li B, Mao J, Wu J, et al. Nano-Bio interactions: biofilm-targeted antibacterial nanomaterials. Small. 2024;20(7):e2306135. https://doi.org/ 10.1002/smll.202306135 DOI: https://doi.org/10.1002/smll.202306135
do Carmo PHF, Garcia MT, Figueiredo-Godoi LMA, Lage ACP, Silva NSd, Junqueira JC. Metal nanoparticles to combat Candida albicans infections: an update. Microorganisms. 2023;11(1):e138. https://doi.org/10.3390/microorganisms11010138 DOI: https://doi.org/10.3390/microorganisms11010138
Kumar D, Kumar A. High throughput bioanalytical techniques for elucidation of Candida albicans biofilm architecture and metabolome. Rend Lincei Sci Fisiche Natu. 2023;34(1):117–129. https://doi.org /10.1007/s12210-022-01115-3 DOI: https://doi.org/10.1007/s12210-022-01115-3
Tscherner M, Giessen TW, Markey L, Kumamoto CA, Silver PA. A synthetic system that senses Candida albicans and inhibits virulence factors. ACS Synth Biol. 2019;8(2):434–444. https://doi.org/10.1021/acssynbio.8b00457 DOI: https://doi.org/10.1021/acssynbio.8b00457
Austermeier S, Kasper L, Westman J, Gresnigt MS. I want to break free–macrophage strategies to recognize and kill Candida albicans, and fungal counter-strategies to escape. Curr Opinion Microbiol. 2020;58:15–23. https://doi.org/10.1016/j.mib.2020.05.007 DOI: https://doi.org/10.1016/j.mib.2020.05.007
König A, Hube B, Kasper L. The dual function of the fungal toxin candidalysin during Candida albicans—macrophage interaction and virulence. Toxins. 2020;12(8):e469. https://doi.org/10.3390/toxins12080469 DOI: https://doi.org/10.3390/toxins12080469
Williams TJ, Harvey S, Armstrong-James D. Immunotherapeutic approaches for fungal infections. Curr Opinion Microbiol. 2020;58:130–137. https://doi.org/10.1016/j.mib.2020.09.007 DOI: https://doi.org/10.1016/j.mib.2020.09.007
Esher SK, Fidel PL, Noverr MC. Candida/staphylococcal polymicrobial intra-abdominal infection: pathogenesis and perspectives for a novel form of trained innate immunity. J Fungi. 2019;5(2):e37. https://doi.org/10.3390/jof5020037 DOI: https://doi.org/10.3390/jof5020037
Van De Veerdonk FL, Netea MG, Joosten LA, Van Der Meer JW, Kullberg BJ. Novel strategies for the prevention and treatment of Candida infections: he potential of immunotherapy. FEMS Microbiol Rev. 2010;34(6):1063–1075. https://doi.org /10.1111/j.1574-6976.2010.00232.x DOI: https://doi.org/10.1111/j.1574-6976.2010.00232.x
d'Enfert C, Kaune A-K, Alaban L-R, et al. The impact of the Fungus-Host-Microbiota interplay upon Candida albicans infections: current knowledge and new perspectives. FEMS Microbiol Rev. 2021;45(3):efuaa060. https://doi.org/10.1093/femsre/fuaa060 DOI: https://doi.org/10.1093/femsre/fuaa060
Zhao S, Shang A, Guo M, Shen L, Han Y, Huang X. The advances in the regulation of immune microenvironment by Candida albicans and macrophage cross-talk. Front Microbiol. 2022;13:e1029966. https://doi.org/10.3389/fmicb.2022.1029966 DOI: https://doi.org/10.3389/fmicb.2022.1029966
Kunyeit L, Anu-Appaiah KA, Rao RP. Application of probiotic yeasts on Candida species associated infection. J Fungi. 2020;6(4):e189. https://doi.org/10.3390/jof6040189 DOI: https://doi.org/10.3390/jof6040189
Allonsius CN, Vandenheuvel D, Oerlemans EFM, et al. Inhibition of Candida albicans morphogenesis by chitinase from Lactobacillus rhamnosus GG. Sci Rep. 2019;9:e2900. https://doi.org/10.1038/s41598-019-39625-0 DOI: https://doi.org/10.1038/s41598-019-39625-0
Copyright (c) 2025 Abdullah, Muhammad Taimur Khan, Zamarak Khan, Hajira Iqbal, Syeda Mahnoor Bukhari, Himmat Khan, Sajjad Ahmad, Muhammad Salman

This work is licensed under a Creative Commons Attribution 4.0 International License.
BSR follows an open-access publishing policy and full text of all published articles is available free, immediately upon publication of an issue. The journal’s contents are published and distributed under the terms of the Creative Commons Attribution 4.0 International (CC-BY 4.0) license. Thus, the work submitted to the journal implies that it is original, unpublished work of the authors (neither published previously nor accepted/under consideration for publication elsewhere). On acceptance of a manuscript for publication, a corresponding author on the behalf of all co-authors of the manuscript will sign and submit a completed the Copyright and Author Consent Form.














