Cataloguing Bacterial Endophytes of Calotropis procera against Potato Early Blight
Abstract
 Abstract Views: 131
Abstract Views: 131
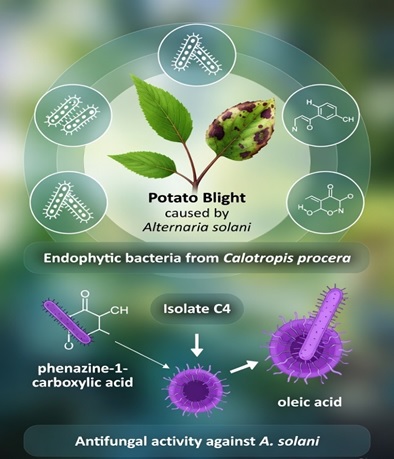
Background. Potato (Solanum tuberosum L.) is a vital food crop, worldwide. It is known for its high nutritious and economic significance, for being a cash and staple crop. However, in Pakistan, its production is seriously hampered by an early blight of potato caused by Alternaria solani, leading to excessive use of fungicides for its control. The current study was conducted to isolate and characterize the endophytic bacteria from Calotropis procera and to identify the secondary metabolites produced by endophytes with the highest antagonism against A. solani.
Method. C. procera samples were collected from the fields of MNS University of Agriculture Multan (MNSUAM). The endophytes were isolated from the leaves and roots of C. procera, followed by their morphological and biochemical characterization. The potato early blight samples were collected from the fields of MNSUAM and A. solani isolation was confirmed after their morphological identification. The 8 isolated endophytes were evaluated for their antagonism against potato early blight pathogen through dual culture assay. The best performing isolate was subjected to gas chromatography-mass spectrometry (GC-MS) based secondary metabolites profiling.
Results. A total of 8 endophytic isolates were evaluated. Among the tested isolates, C4 exhibited the highest antifungal activity (~70%), significantly inhibiting the growth of A. solani. Endophyte C4 produced potent antimicrobial metabolites that effectively suppressed pathogen growth. Based on GC-MS analysis, the major compounds identified were phenazine-1-carboxylic acid (antibiotic, anti-biofilm), oleic acid (membrane disruptor), and others.
Conclusion. The study demonstrated that endophytic bacteria isolated from C. procera, particularly isolate C4, possessed strong antagonistic activity against A. solani. The production of diverse antifungal metabolites highlights their potential as eco-friendly biocontrol agents. These findings support the evaluation of selected endophytes under greenhouse and field conditions for sustainable management of early blight in potato.
Downloads
References
Ghimire E, Karki N, Kafle S, Maharjan SK. Evaluation of potato (Solanum tuberosum L.) planting techniques in high hills of Nepal. J Environ Agric Sci. 2024;26(1&2):11-19.
Swamy KRM. Origin, domestication, taxonomy, botanical description, genetics and cytogenetics, genetic diversity and breeding of potato (Solanum tuberosum L.). Int J Curr Res. 2023;15(4):24352-24372. https:// doi.org/10.24941/ijcr.45099.04.2023
Irfan MF, Fatima N, Ali F, Haider MZ. Assessing potato cultivation techniques in Pakistan: an analysis of existing methods and identified gaps. Bull Biol Allied Sci Res. 2024;2024(1):80-80. https://doi.org/10.54112/bbasr.v2024i1.80 DOI: https://doi.org/10.54112/bbasr.v2024i1.80
Abid S, Nasir J, Anwar MZ, Zahid S. Exponential growth model for forecasting of area and production of potato crop in Pakistan. Pak J Agric Res. 2018;31(1):24-28. https://doi.org/10.17582/journal.pjar/2018/31.1.24.28 DOI: https://doi.org/10.17582/journal.pjar/2018/31.1.24.28
Camire ME, Kubow S, Donnelly DJ. Potatoes and human health. Crit Rev Food Sci Nutr. 2009;49(10):823-840.
https://doi.org/10.1080/10408390903041996 DOI: https://doi.org/10.1080/10408390903041996
Elsharif AA, Dheir IM, Mettleq ASA, Abu Naser SS. Potato classification using deep learning. Int J Acad Pedagog Res. 2020;3(12):1-8.
Oshunsanya SO, Nwosu NJ, Li Y. Abiotic stress in agricultural crops under climatic conditions. In: Jhariya M, Banerjee A, Meena R, Yadav D, editors. Sustainable Agriculture, Forest and Environmental Management. Singapore: Springer; 2019:71-100. https://doi.org/10.1007/978-981-13-6830-1_3 DOI: https://doi.org/10.1007/978-981-13-6830-1_3
Chakrabarti SK, Sharma S, Shah MA. Potato pests and diseases: a global perspective. In: Chakrabarti SK, Sharma S, Shah MA, eds. Sustainable Management of Potato Pests and Diseases. Singapore: Springer; 2022:1-23. https://doi.org/10.1007/978-981-16-7695-6_1 DOI: https://doi.org/10.1007/978-981-16-7695-6_1
Lahlali R, Gachara G, Özer G, Touseef H. Editorial: perspective challenges for applied research in potato pathogens: from molecular biology to bioinformatics. Front Microbiol. 2023;14:e1140107.
https://doi.org/10.3389/fmicb.2023.1140107 DOI: https://doi.org/10.3389/fmicb.2023.1140107
Kumar S, Chandra R. Integrated field application of T. viride, botanicals, and fungicides for managing early blight (Alternaria solani) and enhancement of plant growth, tuber nutritional quality, and potato yield. J Nat Pest Res. 2024;8:e100070. https://doi.org/10.1016/j.napere.2024.100070 DOI: https://doi.org/10.1016/j.napere.2024.100070
Schmey T, Tominello-Ramirez CS, Brune C, et al. Alternaria diseases on potato and tomato. Mol Plant Pathol. 2024;25(3):e13435. https://doi.org/10.1111/mpp.13435 DOI: https://doi.org/10.1111/mpp.13435
Wang X, An K, Guo Y, et al. Uptake, translocation, and subcellular distribution of strobilurin fungicides in cucumber (Cucumis sativa L.). J Agric Food Chem. 2023;71(49):19324-19332. https://doi.org/10.1021/acs.jafc.3c04902 DOI: https://doi.org/10.1021/acs.jafc.3c04902
Mohanan A, Nigam R, Yeliya P, et al. The role of plant microbiomes in suppressing soilborne pathogens: a review. J Adv Microbiol. 2025;25(5):160-178. https://doi.org/10.9734/jamb/2025/v25i5942 DOI: https://doi.org/10.9734/jamb/2025/v25i5942
Kuźniar A, Kruczyńska A, Włodarczyk K, et al. Endophytes as permanent or temporal inhabitants of different ecological niches in sustainable agriculture. Appl Sci. 2025;15(3):e1253. https://doi.org/10.3390/app15031253 DOI: https://doi.org/10.3390/app15031253
Kamran M, Imran QM, Ahmed MB. Endophyte mediated stress tolerance in plants: a sustainable strategy to enhance resilience and assist crop improvement. Cells. 2022;11(20):e3292.https://doi.org/10.3390/cells11203292 DOI: https://doi.org/10.3390/cells11203292
Chowdhury FT, Zaman NR, Islam MR, et al. Anti-fungal secondary metabolites and hydrolytic enzymes from rhizospheric bacteria in crop protection: a review. J Bangladesh Acad Sci. 2020;44(2):69-84.
https://doi.org/10.3329/jbas.v44i2.51452 DOI: https://doi.org/10.3329/jbas.v44i2.51452
Wu X, Lyu Y, Ren H, et al. Degradation of oxalic acid by Trichoderma afroharzianum and its correlation with cell wall degrading enzymes in antagonizing Botrytis cinerea. J Appl Microbiol. 2022;133(5):2680-2693.
https://doi.org/10.1111/jam.15617 DOI: https://doi.org/10.1111/jam.15617
Muthukumar A, Raj TS, Prabhukarthikeyan SR, et al. Pseudomonas and Bacillus: a biological tool for crop protection. In: Singh HB, Vaishnav A, eds. New and Future Developments in Microbial Biotechnology and Bioengineering. Elsevier; 2022:145-158. https://doi.org/10.1016/B978-0-323-85577-8.00006-8 DOI: https://doi.org/10.1016/B978-0-323-85577-8.00006-8
Eid AM, Fouda A, Abdel-Rahman MA, et al. Harnessing bacterial endophytes for promotion of plant growth and biotechnological applications: an overview. Plants. 2021;10(5):e935. https://doi.org/10.3390/plants10050935 DOI: https://doi.org/10.3390/plants10050935
Nofal AM, Hamouda RA, Rizk A, et al. Polyphenols-rich extract of Calotropis procera alone and in combination with Trichoderma culture filtrate for biocontrol of cantaloupe wilt and root rot fungi. Molecules. 2024;29(1):e139.
https://doi.org/10.3390/molecules29010139 DOI: https://doi.org/10.3390/molecules29010139
Baset FM, Hagaggi NS, Hezayen FF, Abdul-Raouf UM. Endophytic bacterial communities colonizing the medicinal plant Calotropis procera: as resources of hydrolases. Nov Res Microbiol J. 2020;4(6):1045-1056.
https://doi.org/10.21608/nrmj.2020.130852 DOI: https://doi.org/10.21608/nrmj.2020.130852
Duhan P, Bansal P, Rani S. Isolation, identification and characterization of endophytic bacteria from medicinal plant Tinospora cordifolia. S Afr J Bot. 2020;134:43-49. https://doi.org/10.1016/j.sajb.2020.01.047 DOI: https://doi.org/10.1016/j.sajb.2020.01.047
Yadav G, Meena M. Bioprospecting of endophytes in medicinal plants of Thar Desert: an attractive resource for biopharmaceuticals. Biotechnol Rep. 2021;30:e00629. https://doi.org/10.1016/j.btre.2021.e00629 DOI: https://doi.org/10.1016/j.btre.2021.e00629
Simionato AS, Navarro MO, de Jesus ML, et al. The effect of phenazine-1-carboxylic acid on mycelial growth of Botrytis cinerea produced by Pseudomonas aeruginosa LV strain. Front Microbiol. 2017;8:e1102.
https://doi.org/10.3389/fmicb.2017.01102 DOI: https://doi.org/10.3389/fmicb.2017.01102
Verma A, Shameem N, Jatav HS, et al. Fungal endophytes to combat biotic and abiotic stresses for climate-smart and sustainable agriculture. Front Plant Sci. 2022;13:e953836. https://doi.org/10.3389/fpls.2022.953836 DOI: https://doi.org/10.3389/fpls.2022.953836
Abbas MM, Ismael WH, Mahfouz AY, et al. Efficacy of endophytic bacteria as promising inducers for enhancing the immune responses in tomato plants and managing Rhizoctonia root-rot disease. Sci Rep. 2024;14:e1331. https://doi.org/10.1038/s41598-023-51000-8 DOI: https://doi.org/10.1038/s41598-023-51000-8
Bahmani K, Hasanzadeh N, Harighi B, Marefat A. Isolation and identification of endophytic bacteria from potato tissues and their effects as biological control agents against bacterial wilt. Physiol Molecul Plant Pathol. 2021;116:e101692. https://doi.org/10.1016/j.pmpp.2021.101692 DOI: https://doi.org/10.1016/j.pmpp.2021.101692
Liu Y, Zhou Y, Qiao J, et al. Phenazine-1-carboxylic acid produced by Pseudomonas chlororaphis YL-1 is effective against Acidovorax citrulli. Microorganisms. 2021;9(10):e2012.
https://doi.org/10.3390/microorganisms9102012 DOI: https://doi.org/10.3390/microorganisms9102012
Faiq M, Ali A, Shafique S, et al. Endophytic fungi as biocontrol agents: a metabolite-driven approach to crop protection and sustainable agriculture. Physiol Mol Plant Pathol. 2025;140:e102857.
https://doi.org/10.1016/j.pmpp.2025.102857 DOI: https://doi.org/10.1016/j.pmpp.2025.102857
Huang W. Recent advances in phenazine natural products. Int J Mol Sci. 2024;25(18):e10345. https://doi.org/10.3390/ijms251810345
Copyright (c) 2025 Hasan Riaz, Abdul Hadi, Muhammad Shahzad Zafar

This work is licensed under a Creative Commons Attribution 4.0 International License.
BSR follows an open-access publishing policy and full text of all published articles is available free, immediately upon publication of an issue. The journal’s contents are published and distributed under the terms of the Creative Commons Attribution 4.0 International (CC-BY 4.0) license. Thus, the work submitted to the journal implies that it is original, unpublished work of the authors (neither published previously nor accepted/under consideration for publication elsewhere). On acceptance of a manuscript for publication, a corresponding author on the behalf of all co-authors of the manuscript will sign and submit a completed the Copyright and Author Consent Form.














