Development and Validation of a UV-Spectrophotometric Method for the Estimation of Atorvastatin in Bulk and Tablet Dosage Form
Abstract
 Abstract Views: 70
Abstract Views: 70
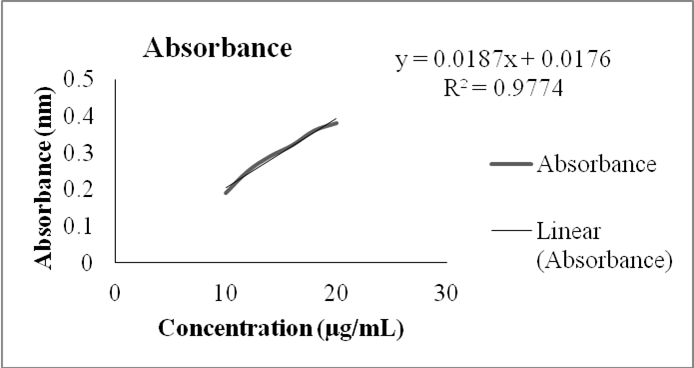
A new UV-spectrophotometric method is devised that uses methanol as a solvent to precisely measure the quantity of atorvastatin present in both raw materials and pharmaceutical dosage form. It is efficient, accurate, simple, quick, and distinctive. High sensitivity and accuracy are the distinguishing features of this approach. Atorvastatin’s max was located at 247 nm. A linear relationship between the concentrations of 10-20 g ml-1 was discovered, exhibiting a high correlation coefficient. The new method’s precision, linearity, accuracy, and limit of detection (LOD) were all validated statistically. The results affirm the suitability of this method for daily analysis of atorvastatin in both its raw form and pharmaceutical formulations. Furthermore, it is noteworthy that this method avoids the need for costly solvents, extraction procedures, derivatization, and time-consuming steps. Hence, it is successfully applied to pharmaceutical formulations and validated in harmony with the standard procedures of the International Council of Harmonization (ICH)
Downloads
References
Shitara Y, Sugiyama Y. Pharmacokinetic and pharmacodynamic alterations of 3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase inhibitors: drug-drug interactions and interindividual differences in transporter and metabolic enzyme functions. Pharmacol. Ther. 2006;112(1):71-105. https://doi.org/10.1016/j.pharmthera.2006.03.003
Bakker-Arkema RG, Davidson MH, Goldstein RJ, et al. Efficacy and safety of a new HMG-CoA reductase inhibitor, atorvastatin, in patients with hypertriglyceridemia. J Am Med Assoc. 1996;275(2):128-133.
Nawrocki JW, Weiss SR, Davidson MH, et al. Reduction of LDL cholesterol by 25% to 60% in patients with primary hypercholesterolemia by atorvastatin, a new HMG-CoA reductase inhibitor. Arterioscler Thromb Vasc Biol. 1995;15(5):678-682. https://doi.org/10.1161/01.atv.15.5.678
Jamshid A, Nateghi AR. HPTLC determination of atorvastatin in plasma. Chromatographia. 2007;65(11):763-766. http://dx.doi.org/10.1365/s10337-007-0228-4
Al-Shehri MM. A validated capillary electrophoresis method for simultaneous determination of ezetimibe and atorvastatin in pharmaceutical formulations. Saudi Pharma J. 2012;20(2):143-148. https://doi.org/10.1016%2Fj.jsps.2011.09.005
Erk N. Development of electrochemical methods for determination of atorvastatin and analytical application to pharmaceutical products and spiked human plasma. Crit Rev Anal Chem. 2004;34(1):1-7. http://dx.doi.org/10.1080/10408340490273717
Korany MA, Hewala II, Abdel-Hay KM. Determination of etofibrate, fenofibrate, and atorvastatin in pharmaceutical preparations and plasma using differential pulse polarographic and square wave voltammetric techniques. J AOAC Int. 2008;91(5):1051-1058. https://doi.org/10.1093/jaoac/91.5.1051
Sharaf-E-Din MMK, Salama FMM, Nassar MWI, Attia KAL, Kaddah MMY. Validated spectrofluorimetric method for the determination of atorvastatinin pharmaceutical preparations. J Pharma Anal. 2012;2(3):200-205. https://doi.org/10.1016/j.jpha.2012.01.005
Erturk S, Aktas ES, Ersoy L, Ficicioglulu S. An HPLC method for the determination of atorvastatin and its impurities in bulk drug and tablets. J Pharm Biomed. Anal. 2003;33(5):1017-1023. https://doi.org/10.1016/s0731-7085(03)00408-4
Petkovska R, Cornett C, Dimitrovska A. Development and validation of rapid resolution RP-HPLC method for simultaneous determination of AT and related compounds by use of chemometrics. Anal Lett. 2008;41(6):992-1009. http://dx.doi.org/10.1080/00032710801978566
Rajeswari KR, Sankar GG, Rao AL, Seshagirirao JVLN. RP-HPLC method for simultaneous determination of atorvastatin and amlodipine in tablet dosage form. Indian J Pharm Sci. 2006;68(2):275-277. http://dx.doi.org/10.4103/0250-474X.25738
Sonawane SS, Shirkhedkar AA, Fursule RA, Surana SJ. Application of UV-spectrophotometry and RP-HPLC for simultaneous determination of atorvastatin calcium and ezetimibe in pharmaceutical dosage form. Eurasian J Ana Chem. 2006;1(1):31-41. http://dx.doi.org/10.12973/ejac/77004
Bhinge SD, Malipatil SM. Development and validation of a stability-indicating method for the simultaneous estimation of cefixime and dicloxacillin using the RP-HPLC method. J Taibah Uni Sci. 2016;10(5):734-744. http://dx.doi.org/10.1016/j.jtusci.2015.10.011
Almutairi FM, Althobaiti YS, Abduljabbar MH, et al. A green and sustainable UV spectrophotometric approach for simultaneous determination of rosuvastatin, pravastatin, and atorvastatin in pharmaceuticals leveraging firefly algorithm-enhanced partial least squares regression. Anal Methods. 2025;17(20):2485-2494. https://doi.org/10.1039/D5AY00446B
Kumari D, Syed SM. Precise and accurate UV spectrophotometric estimation of atorvastatin in bulk and dosage form: a validation study. Int J Pharm Sci Res. 2025;16(12):3421-3425. https://doi.org/10.13040/IJPSR.0975-8232.16(12).3421-25
Copyright (c) 2026 Muhammad Aslam, Fahad Mushtaq, Habib Raza, Zahra Noreen, Aamir Sohail, Muhammad Aneeq Javed, Mehvish Abdul-Rehman

This work is licensed under a Creative Commons Attribution 4.0 International License.






